By Dave Ongie, Managing Editor
As cases of COVID-19 surge in our region, Ballad Health opened a monoclonal antibody infusion center in North Johnson City last week to help treat patients who are infected with the virus.
The center is located at 378 Marketplace Blvd. Suite 5, and treatments are available by appointment only. Ballad Health Chief Operating Officer Eric Deaton said the 12-bed treatment center is helping keep folks out of the hospital during the current surge.
“We’ve been using that since it first received FDA emergency use authorization back in December of 2020,” Deaton said during a media briefing last Thursday. “It’s showing tremendous promise and great results with our patients.”
Dr. Mark Wilkinson, Ballad’s chief medical information officer, said the antibody treatments work in a very similar manner to COVID-19 vaccines, targeting spike proteins to initiate an immune response that helps a person fight off the virus.
“The monoclonal antibodies have been achieved by extracting that antibody from somebody who has had COVID-19, replicating it and then making it available as a drug to give to patients who suffer from COVID-19,” Wilkinson said. “In essence, that jumpstarts the immune response so those antibodies get into the bloodstream immediately to start fighting off that infection.”
Treatments are most effective in patients within the first 7-10 days of infection, and there are some qualifications in order to receive treatment. For starters, patients must be over the age of 12 and weigh at least 88 pounds. If patients are under the age of 65, they must have a risk factor, such as heart, lung or kidney disease in order to receive treatment. Pregnant women also qualify for the treatment.
The treatment is usually administered through an IV valve for 20 minutes, but can also be given in four separate shots. Patients must be observed for an hour after treatment to ensure there is no adverse reaction.
Wilkinson said patients who receive the treatment quickly after contracting COVID-19 are seeing quick relief in most cases.
“People tend to feel better in the next 24 to 48 hours depending upon how bad you felt to begin with, how severe your disease was and how you responded to the therapy itself,” Wilkinson said.
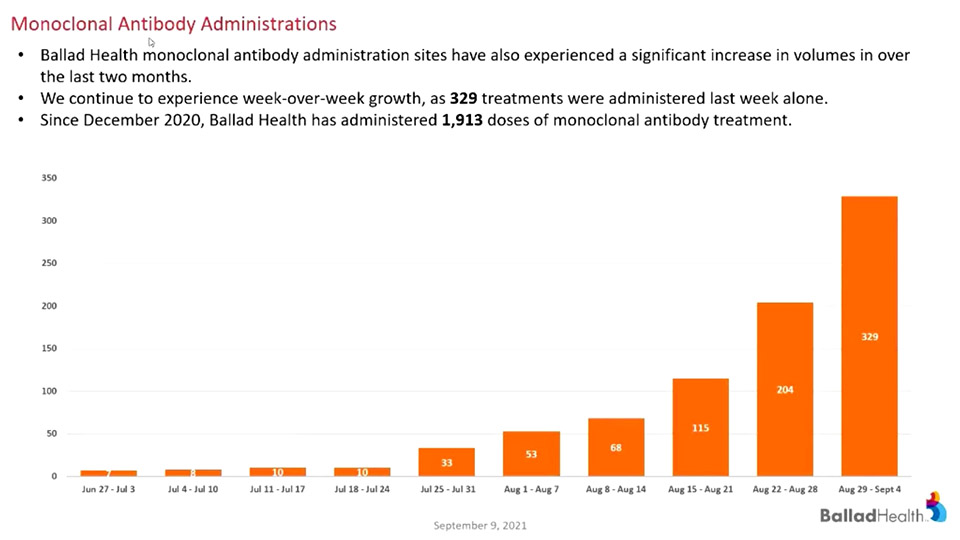
In recent weeks, the demand for the treatment has risen exponentially as the delta variant continues to spread through our region.
“To put this in perspective, we’ve administered more doses in the past six weeks due to the surge in COVID-19 than we did in the first six months it was available to us,” Wilkinson said.
As promising as the treatment has been, Deaton and Wilkinson both urge anyone who has not already been vaccinated to get vaccinated.
“We’d always prefer folks get vaccinated, that way they may never need this therapy: Wilkinson said.



